
You can also get the formula used in Chlorine (Cl) - Standard Atomic Weight to Table salt (sodium chloride) (NaCl) - Molecular Mass conversion along with a table representing the entire conversion. If you encounter any issues to convert, this tool is the answer that gives you the exact conversion of units. Converting Chlorine (Cl) - Standard Atomic Weight to Table salt (sodium chloride) (NaCl) - Molecular Mass is easy, for you only have to select the units first and the value you want to convert. (note the unit - g per mole or g.mole(-1)) 1 mole of Cl35 contains an Avagadro’s number of atoms, N where N 6.023 x 1023 per mole (n. When you are converting molar mass, you need a Chlorine (Cl) - Standard Atomic Weights to Table salt (sodium chloride) (NaCl) - Molecular Masss converter that is elaborate and still easy to use. Answer (1 of 2): Chlorine has two common isotopes: Cl35 and Cl37 Select one - say Cl35 1 mole of Cl35 has a mass, m, of 35.00 g / mole by definition. to NaCl M.W through multiplicative conversion factors. helps in the conversion of different units of measurement like Cl Std. Be it buying grocery or cooking, units play a vital role in our daily life and hence their conversions. M A v g ( Natural Abundance 100 × M molar Mass) Here summation is applied on two isotopes of chlorine. 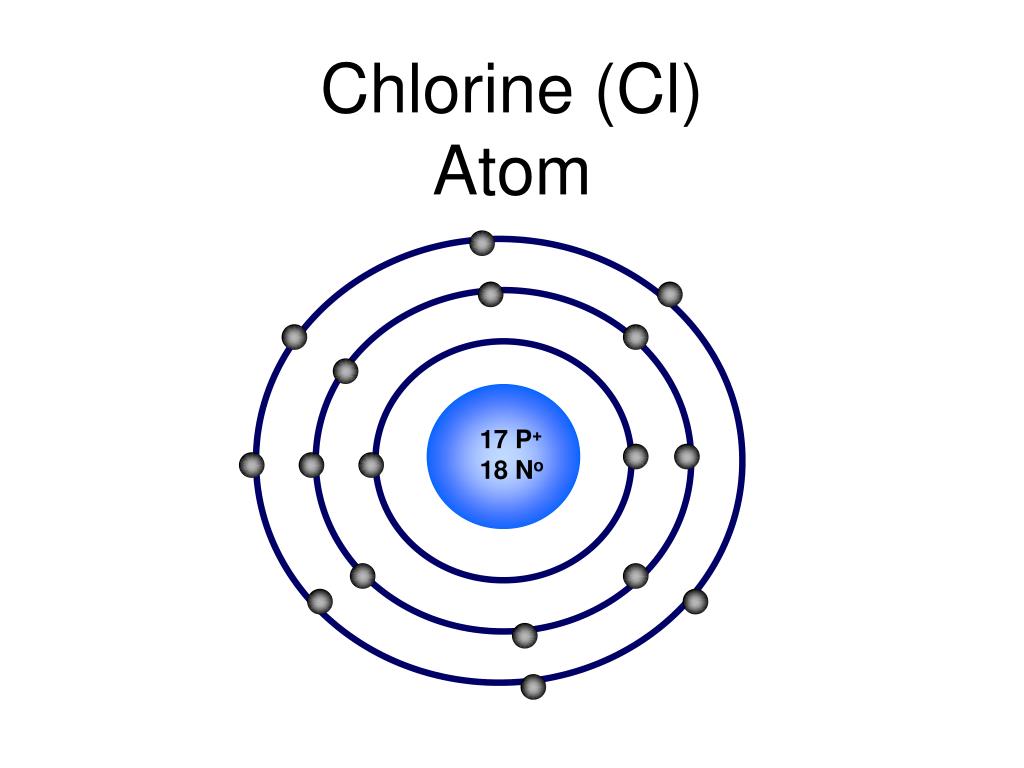
Average atomic mass of chlorine is given by the sum of the product of abundance ( p e r c e n t a g e / 100) and molar mass for all isotopes. Measurement like molar mass finds its use in a number of places right from education to industrial usage. Molar masses of 35 C l and 37 C l are 34.9689 and 36.9659 respectively. Units of measurement use the International System of Units, better known as SI units, which provide a standard for measuring the physical properties of matter. Chlorine (Cl) - Standard Atomic Weights to Table salt (sodium chloride) (NaCl) - Molecular Masss Converter
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
